Digital platform launch for CEED of Charité Berlin to promote MedTech innovations in the healthcare sector
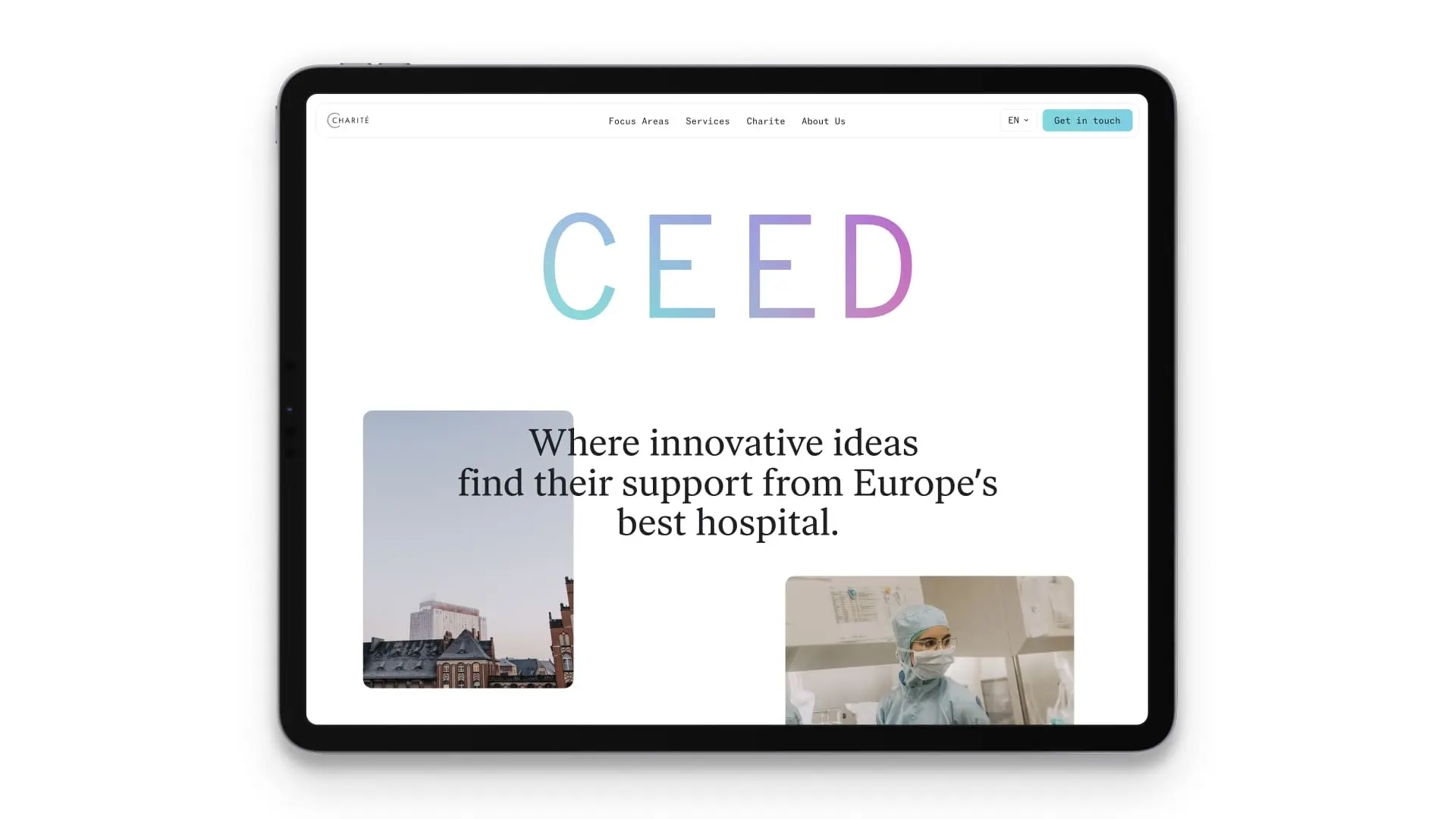
The CEED platform revolutionizes the way industry and clinic work together in the MedTech industry. The industry-in-clinic platform offers the industry unique access to Charité's excellent clinical expertise, consultations around complex regulatory requirements and iterative evaluations of ideas, prototypes and products.
Professor Thomas Picht, MD, Director of Division CEED & Chief Medical Officer, stated, "With CEED, we aim to foster innovation in healthcare by bridging the gap between the MedTech industry and clinical application. We are excited to shape the medicine of tomorrow and thank the great team at Forte Digital for the fantastic collaboration."
Increased effectiveness and rapid integration into standard care
The collaboration between CEED and Forte Digital has produced an impressive and effective platform in a very short time. Joachim Bader, Managing Partner at Forte Digital Central Europe, underlined the achievement: "Within six months, we were not only able to develop a brand with the Charité team, but with the conception and launch of a digital platform, we were also able to bring CEED directly to life and thus help the CEED team and companies quickly come together. This is exactly the kind of assignment we love, where brand, product, user experience and business impact are in demand."
By using agile methods of iterative refinement and evaluation with different users, the platform accelerates product development cycles and promotes demand-driven developments.
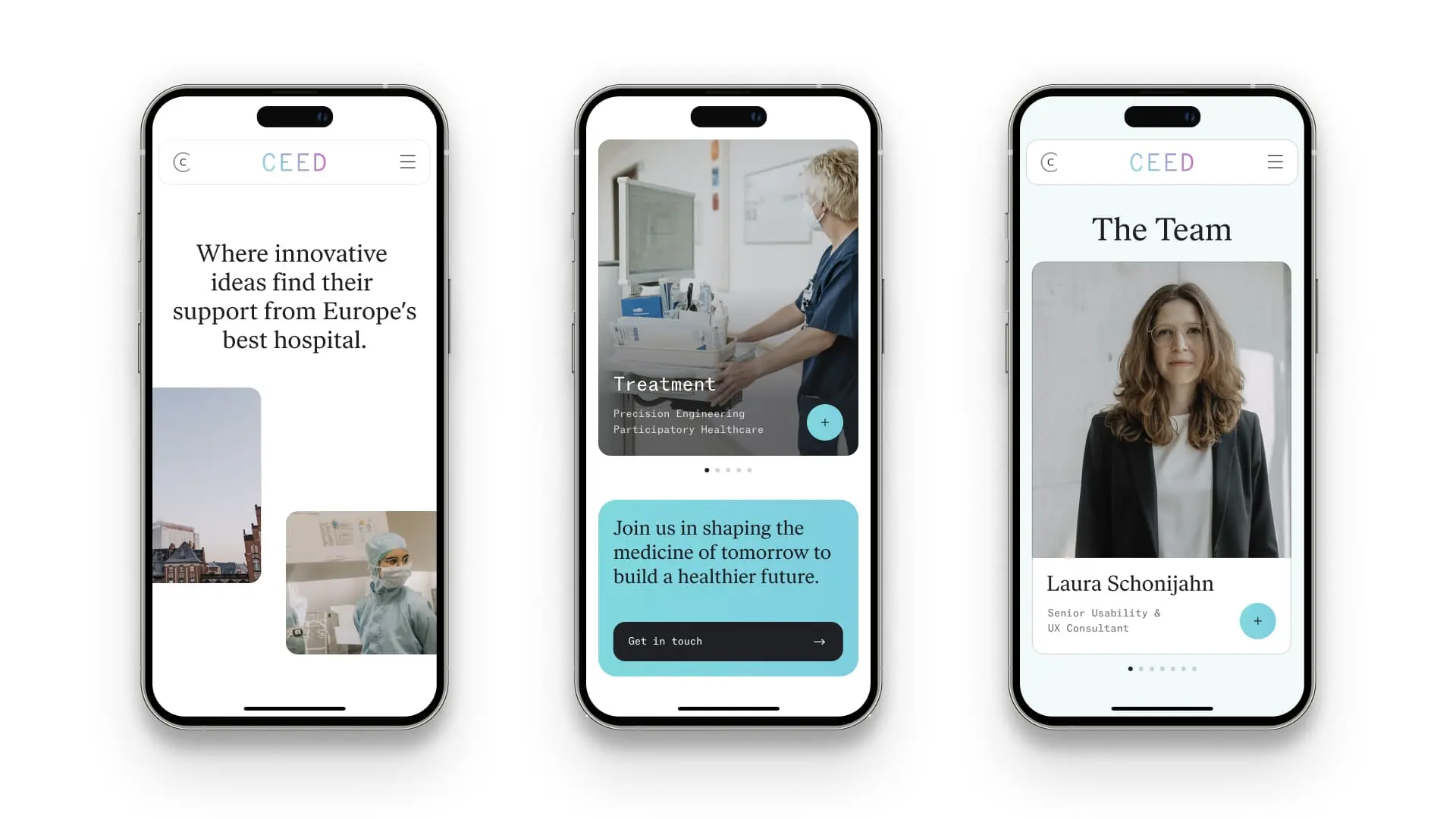
Tailor-made innovation: versatile business areas to promote MedTech development
CEED's three business areas include consulting, evaluation and education. Each customer receives a tailored plan to drive product developments based on user-oriented research and agile project management. The building blocks range from needs assessment and concept development to consulting services on product approval and other regulatory frameworks to (pre-) clinical evaluation and education strategy. CEED also supports analysis and consulting on interoperability issues, implementability in IT infrastructures, hospital processes and the development of educational programs.
Joint forces for a transformative MedTech future
Forte Digital helped CEED create a strong brand identity and design and implement the website. The partnership between CEED and Forte Digital embodies the interplay of expertise and passion that is reshaping the future of the MedTech industry.
Fotos © Charité - Janine Oswald
About CEED - The MedTech consulting platform of Charité:
CEED has been an independent business unit of Charité Healthcare Services GmbH, a subsidiary of Charité - Universitätsmedizin Berlin, the largest university hospital in Europe, based in Berlin, since 01.01.2023. With funding from the German Federal Ministry of Education and Research (BMBF), CEED strives to drive innovation in healthcare. Led by Prof. Dr. med. Thomas Picht as Director of the CEED Division, Denny Chakkalakal as Head of Operations and Mirko Jonas as Director of Finance, CEED acts as a link between the MedTech industry and clinical applications. CEED's goal is to bridge the gap between innovative MedTech solutions and their integration into clinical care. By supporting demand-driven developments, CEED helps innovations find their way into mainstream care more quickly. CEED's services include regulatory consulting, clinical insights, market access, and prototype and product evaluation.
Read more:
Contact us:

Nils Hocke
Director Forte Improve Germanynils.hocke@fortedigital.com





